|
So even if the outermost electron of lithium and the outermost electrons of fluorine reside on the same energy level, the latter will be more attracted to the nucleus, since fluorine has #9# protons there.Īs a result, the atomic size of fluorine will be smaller than that of lithium, or, in other words, lithium will have larger atomic radius than fluorine. 
for instance a lithium atom (Z3, A7 amu) contains three protons (from Z), three electrons (as proton electron), and four neutrons (7 3 4). Since outermost electrons are located on the same energy level in both the lithium and the fluorine atoms, it follows that the nucleus that has more protons will attract these electrons more - this is known as effective nuclear charge. Learn more about Atomic Number Mass Number in detail with notes, formulas, properties, uses of Atomic Number Mass Number prepared by subject matter experts. Here is where the number of protons that each atom has in its nucleus becomes important. Now, an atom's size is actually determined by how far away from the nucleus its outermost electrons are located.Ītoms that share a period have one important thing in common - their outermost electrons are located on the second energy level. Periodic table Periodic table of the chemical elements color-coded to show four sets of elements referred to as blocks. So fluorine has more protons in its nucleus. At the left, and including helium (He) at the top right, is the s-block at the foot is the f-block above that, spanning colums 3 to 12, is the d-block and at the right is the p-block (excl. 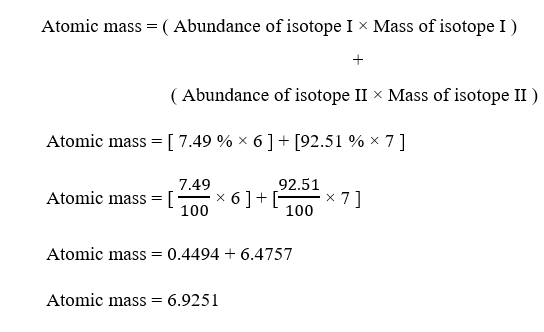
In your case, lithium has an atomic number equal to #3# and fluorine an atomic number equal to #9#. Classification, Properties and Characteristics of Lithium Atomic mass/weight, 6.938 atomic mass units Molar mass/Molecular weight, 6.94 g/mole Mass Number, 7. Periodic table Periodic table of the chemical elements color-coded to show four sets of elements referred to as blocks. So, you know that atomic number, which represents the number of protons that can be found in the nucleus of an atom, increases as you move from left to right across a period. This means that you can use your knowledge of the basic atomic structure to determine which atom will have the larger atomic radius. Notice that lithium is located in group 1 and fluorine is located in group 17 and, more importantly, they are both lcoated in period 2 of the periodic table. So, just like a moving mass has kinetic energy 1/2 mv2, a coil carrying. Start by taking a look at a periodic table and making a note of where lithium, #"Li"#, and fluorine, #"F"#, are located. comAtomic Number of Lithium is 3, so it has 3 electrons in its neutral state.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
